Disclaimer : this article is only intended to provide some background for people planing to use Lithium based batteries. It has been written from the own experience of the authors, and in no way constitutes a safety rule or whatever similar you could think about. You have to inform and educate yourself, using all available material on the topic, and then make your own rules by yourself.
By no mean POBOT association or its members can be made responsible for any damage, injury,... that could occur while using such products.
Lithium type batteries provide a highly attractive capacity/weight ratio, compared to NiCd (Nickel-Cadmium) or NiMH (Nickel-Metal Hybrid) ones. For the same capacity they weight about one third of a "traditional" pack.
Li-Ion & Li-Po batteries overview
Both kinds of batteries are built around Lithium based chemistry. The first one uses LI+ ions in a liquid electrolyte (hence requiring a rigid casing). The second one uses a solid polymer as electrolyte, which allows a supple envelope.
Beware : these batteries are convenient thanks to their small form factor, but it is mandatory to take a minimal care when using or charging them. A wrong usage can have dramatic consequences, such as fire hazard. LiPO ones are especially vulnerable because of their supple envelope which can be pierced.
Several other kinds of Lithium based batteries can be found too. They differ either by an innovation (e.g. nano-particles) such as Lithium Iron Phosphate / A123, or by using different metals for the positive electrode (anode) : Lithium Manganese or Lithium Cobalt. These technologies are detailed in an other article : Les batteries LiFePO4 ou A123
If you don’t find the needed information, feel free to contact us : contact@pobot.org
Advantages
They are lightweight and provide an important capacity in a small volume. They have a self-discharge rate as low as 10% per month, which allows to store ready to use batteries and have them operational at once (as with Pb based batteries). This is not the case for classical (NiMH) ones, which require to be periodically recharged when not in use.
Drawbacks
- They have limits with respect to discharge and charge ratios :
- discharge current cannot exceed 15 times the capacity [1] (for the best ones)
- charge current is limited to 1 time the capacity, thus it takes at least 1 hour to charge an "empty" battery(in practical use, count twice or three times this value).
- They are fragile, both on a mechanical and electrical standpoints.
- They are expensive :( but things are changing :)
Availability
These batteries are widely available nowadays, either as separate elements or as assembled packs with standard connectors.
Technical details and typical charts
They are generally available as 4.2 volts elements, instead of 1.2 volts for traditional batteries.
Charge / discharge characteristics
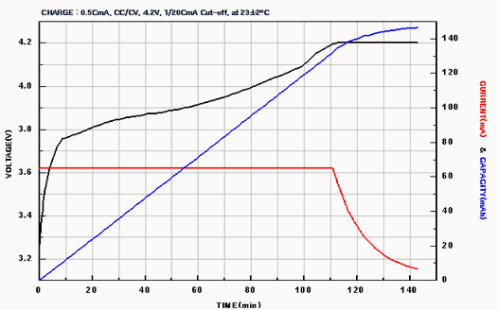
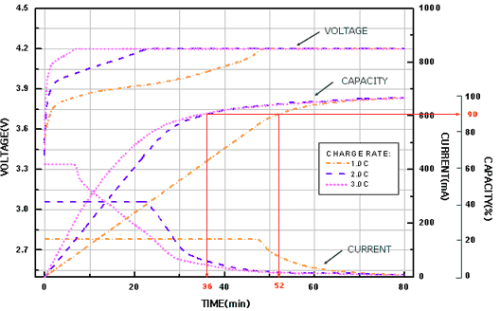
At the beginning of the charge and during the first phase, the current is limited and the voltage increases progressively until reaching the regulated voltage.
Starting from there, we reach the second phase, know as "filling". The voltage stays perfectly stable and the current decreases.
The charge is complete when the current goes under the 0.03C threshold. It is then mandatory to stop the process, since Lithium batteries don’t tolerate being overcharged at all.
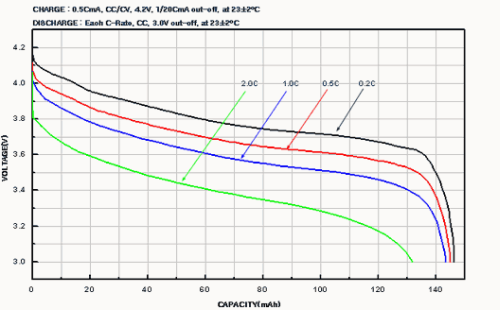
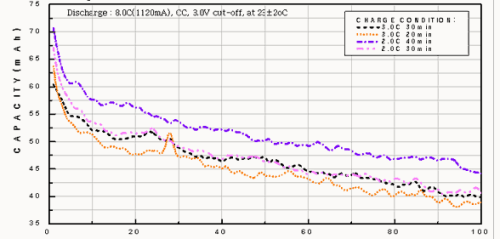
Capacity vs. discharge characteristics
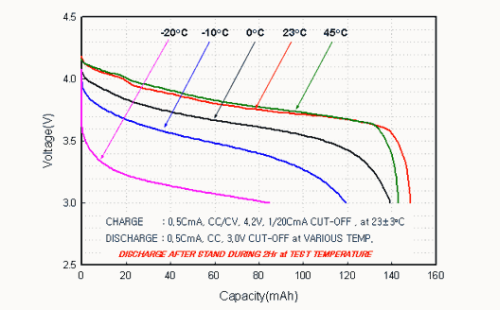
Capacity vs. ambient temperature characteristics
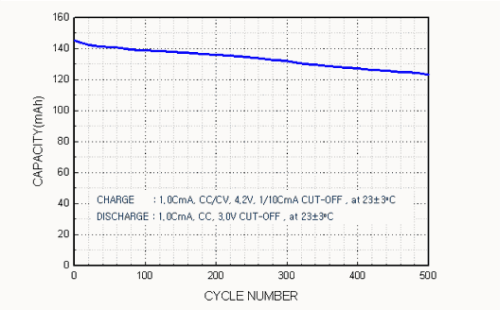
Capacity vs. charge/discharge cycle count characteristics
Charge delay vs. charge current (A))
Delivered capacity vs. charge ratio
Safety rules and good practices
Hereafter rules can appear as overstraining, but they must be followed, and by the way they are more or less valid for any kind of battery.
NiCd ones are maybe more tolerant to bad treatments, but they suffer from them and their performances decrease quite rapidly then. A lot of accidents have also occurred with NiCd batteries, a model maker having had his hand torn off by one : under certain conditions, NiCd elements can explode like grenades, while LiPo only catch fire (which is quite dangerous too anyway :) )
A few charging rules
- Before any recharge or use, always check the voltage on pack terminals !!
- Once the charge is completed, never try to start a new charge cycle, in the intend to fill them at the most. The charger will then force a high current when starting the cycle, which will lead to overheating of the element(s), and thus to fire hazard.
- Only specifically designed chargers must be used for charging LiPo elements, since they are the only ones ensuring to respect of constraints and charge profiles.
- Use only reliable connectors to avoid shorting elements by inadvertence. Female terminals are recommended on pack side, male ones on charger side.
- Take special care with chargers using banana terminals. Always disconnect packs from leads before any manipulation of the terminals connected to the charger. It is very easy to short this kind of terminals, since both positive (red) and negative (black) ones are male.
- Use CC/CV (constant current / constant voltage) chargers for best results. Such models use 1C charge current and 4.2V voltage, and decrease the charge rate when approaching the end of the process.
- Elements are charged up to 90% after 1 hour, and can be left connected for 40 to 50 minutes after that to reach a full charge. The maximum allowed charge voltage is 4.25V per element. The maximum allowed charge current is 1C (f.i. 700mA for a 700mAh element)
- In no case should you try to charge LiPo elements with a current above 1C. This would shorten the element life and would provide a very limited time saving. Always prefer a slow charge.
- There is no point trying to increase charging current in the intent to shorten the charge time. If it allows reaching the transition between the first and second phases sooner, the filling phase will take longer. Moreover, increasing the current has a direct consequence on the lifetime of the battery. It is thus wiser to limit charging current to C/2.
- Never leave Li-ion or LiPo under standby charge (in the intent to keep them fully charged). This creates a Lithium coating on electrodes, which makes the element unstable. To compensate for self-discharge, it is advised to briefly recharge the battery event 500 hours, this process being stopped when current drops under 0.03C.
- Very important : Charging as a single element a LiPo packs made of several elements connected in serial is not a good idea. Such packs are not equipped with protections such as Li-ion ones, and this can lead to voltage discrepancies between the elements of the pack, because only the global voltage of the pack is regulated by the charger. It is then possible to see some of the elements reaching 4.3 V while others stay at 4.1 V only, the overall pack voltage being still 4.2 V. Individual element voltage must be monitored. This is the role of balancing connectors available on packs and chargers, allowing the regulation circuitry to monitor individual element voltages and not only the pack one. The rule is to always use a balancing charger when charging packs not internally equipped with balancing circuitry.
Charge must be done with a constant voltage and a limited current.
Specificity of Lithium batteries charges can be summarized to two major points :
- They don’t accept overloading nor short-circuit, even the slightest.
- You must observe the charging voltage, otherwise they will not charge.
This have the consequence that the range between the minimal charge voltage and the absolute maximum voltage is very tight. Charging voltage must thus be very precisely regulated.
- Both Li-ion and LiPo technologies have close but different charging voltages :
- Li-ion : 4.1 Volts per element
- Li-po : 4.2 Volts per element
Nowadays accepted tolerance is +/- 0.50V per element only, so it is wise to check you’ve selected the appropriate settings before starting a charge cycle.
- Charging current must be set between C/2 and 1C. With these configuration, it takes between 2 and 3 hours to charge a battery.
- Some chargers claiming 1 hour charge delay for Lithium batteries do in reality a partial charge. After 1 hour, on 75% of the element charge is reached, whatever is the charging current. As said above, it takes time after this point for reaching a full charge.
- If an element is overcharged it produces hydrogen. Overpressure and overheating occur then, which can lead to an explosion. Li-ion packs are generally protected against overheating and overpressure by internal mechanisms. In contrary, supply LiPo packs are sensitive, and even if they don’t explode violently, they can produce huge flames which can cause severe burns.
If ever you notice a temperature raise, this is abnormal : stop immediately.
Precaution of use
A lot of common life objects (cars, knives,...) can create severe damages and injuries if they are used without observing a few elementary safety rules. This is exactly the same for LiPo batteries.
- Safety systems (control circuitry) start to be available. Such systems are embedded in packs and prevent most of the accidents. Prefer these kinds of packs.
- Double check that your charger is correctly configured for the battery you are going to charge.
- Voltage and intensity must be both correct. Always double check this. An overloading is the best way for having the pack catch fire.
- You always must charge elements on a fire-proof surface, ideally a ceramic made box with top side opened. Some people use stone-like rectangular flower pots, other prefer doing this in their chimney. But in no case should you use metal boxes.
- Never charge near flammable liquid or solid material, such as wood furnitures, curtains,...
- New elements have a high initial charge. You must take care that such elements have no chance to be shorted :
- Store them in a fresh and ventilated place
- Never put them in the pocket (keys could short them), or in a bag where some other objects could short them
- Never store them ’bulk’
- Never put them on conductive surfaces : metal box, metal table,...
- Do not expose elements to heat (the car quarter-deck on a sunny day for instance)
- Never leave packs in children reach.
- Ensure packs cannot be pierced by metallic objects (for instance, don’t store them in a toolbox).
- A pack having been damaged by a shock (RC plane crash for instance) is potentially dangerous and can catch fire because of an external shorting.
- It can take more than 10 minutes for a damaged pack to catch fire. Because of this, a shortened pack (event for a very short period) must be placed under watch after the incident.
- In the event some electrolyte comes in contact with skin, you should profusely clean it (the skin, not the pack) with soap and water and consult a doctor shortly.
- Batteries must be fully discharged before being disposed of. A not discharged element can still catch fire after disposal.
- Use only batteries designed for the usage you plan to do.
Discharge
As for other technologies, Lithium batteries have a minimal voltage, under which you must in no way discharge them, at the risk of destroying the elements. The absolute lowest element voltage is 2.5 volts, but under normal use you must not go under 3 volts. Anyway, once the 3 volts level reached, things go faster, since this part of the discharge curve is very steep, and thus the voltage drops faster and faster.
- As already mentioned, the self-discharge of Lithium batteries is around 10% per month.
- Nowadays Lithium batteries are able to sustain high currents. Their internal resistance is now very close to NiCd and NiMh ones :
– internal Li-ion resistance : between 150 and 250 milliOhms
– internal LiPo resistance : between 200 and 300 milliOhms
This allows current as high as 1C, and even 2C. Some special LiPo models are able to sustain 10C currents, and 15C for peaks.
We already said it several times, but this is of the utter importance : never short a Lithium battery.
 PoBot
PoBot
 Li-Ion and Li-PO batteries
Li-Ion and Li-PO batteries



